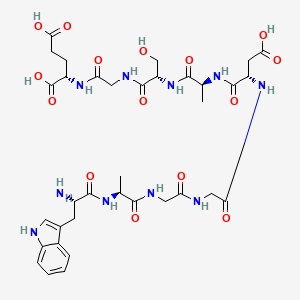
Research Applications
Sleep Regulation and Circadian Rhythm Optimization
DSIP demonstrates remarkable efficacy in promoting deep, restorative sleep without the sedative effects characteristic of traditional sleep medications. Research published in human sleep studies shows that DSIP administration significantly improved sleep efficiency and reduced sleep latency in insomniac patients. A controlled double-blind study found that DSIP treatment resulted in higher sleep efficiency and shorter time to fall asleep compared to placebo, though effects were modest in chronic insomnia cases.
In healthy volunteers, acute DSIP administration produced immediate feelings of sleep pressure and increased total sleep time by 59% within a 130-minute interval following treatment compared to placebo. Importantly, delayed effects on subsequent night sleep included shorter sleep onset, reduced percentage of stage 1 sleep, and improved overall sleep efficiency. Sophisticated behavioral and EEG analyses revealed that DSIP sustains natural sleep functions without causing sedation through classic pharmacological mechanisms.
Animal studies demonstrate that DSIP increases the duration and quality of slow-wave sleep (SWS), which is the deepest and most restorative phase of the sleep cycle. Research in rats showed DSIP increased the percentage of slow-wave sleep by 32% and total sleep time by 72%. The peptide appears to modulate sleep-wake functions with greater activity in circumstances where sleep is disrupted, making it particularly valuable for individuals experiencing stress-related sleep disturbances.
DSIP's effects on circadian rhythms extend beyond immediate sleep induction. The peptide influences the body's natural production of melatonin, the primary circadian rhythm regulator, thereby supporting the synchronization of internal biological clocks with natural light-dark cycles. By enhancing endogenous melatonin secretion rather than providing exogenous supplementation, DSIP offers a more physiologically natural approach to circadian rhythm optimization.
Sources:
Stress Reduction and HPA Axis Modulation
DSIP demonstrates significant stress-protective properties through its modulation of the hypothalamic-pituitary-adrenal (HPA) axis, the body's central stress response system. Research shows that DSIP influences key stress hormones including cortisol, ACTH, and corticosterone, helping to normalize stress-induced metabolic and functional disorders in both human and animal subjects exposed to various stressors.
The peptide's stress-modulating effects appear to work through multiple neurotransmitter systems. DSIP influences gamma-aminobutyric acid (GABA) activity, the brain's primary inhibitory neurotransmitter, promoting feelings of calmness and relaxation. Additionally, the peptide modulates serotonin pathways, which are essential for mood regulation and emotional well-being. By stabilizing these neurochemical systems, DSIP helps the body manage stress responses more effectively.
Clinical studies demonstrate DSIP's capacity to reduce elevated cortisol levels associated with chronic stress. The peptide's regulatory influence on the HPA axis prevents the prolonged elevation of stress hormones that can lead to metabolic dysfunction, immune suppression, and cognitive impairment. This cortisol-lowering effect contributes to improved mental resilience and may be particularly beneficial for individuals experiencing burnout syndrome or stress-related disorders.
Research indicates that DSIP administration produces anxiolytic effects without causing dependency or tolerance. Long-term animal studies showed that DSIP-treated mice spent 73% more time in open arms of elevated maze tests and explored maze extremities 9 times more frequently than controls, indicating reduced anxiety-like behaviors. These effects persisted throughout the animals' lifespans, suggesting sustained anxiolytic benefits without adaptation or diminished response.
The peptide's stress-protective actions extend to prevention of stress-induced physiological damage. Studies show DSIP decreases stress-related metabolic disturbances, reduces inflammatory cytokine production (including IL-6 and TNF-α), and protects against oxidative stress in neural and peripheral tissues. These protective effects contribute to overall stress resilience and may prevent the long-term health consequences of chronic stress exposure.
Sources:
Pain Management and Analgesic Properties
DSIP demonstrates potent analgesic effects for various pain conditions, particularly chronic and neuropathic pain that often proves resistant to conventional treatments. Research shows the peptide significantly elevates pain thresholds through mechanisms distinct from traditional opioid analgesics, as DSIP-induced analgesia is insensitive to naloxone (an opioid receptor antagonist), suggesting alternative pain-modulating pathways.
Animal studies examining circadian pain thresholds found that rats treated with DSIP (0.1 mg/kg) exhibited significantly higher pain threshold levels measured by hot plate testing in both light and dark periods. Treatment with DSIP also shifted the peak pain threshold from 10:00 to 18:00, indicating the peptide's influence on circadian pain modulation. Higher doses (1.0 mg/kg) increased pain thresholds specifically during dark periods, demonstrating dose-dependent and time-specific analgesic effects.
Clinical trials with human patients suffering from chronic pain conditions have shown remarkable therapeutic efficacy. A pilot study involving seven patients with conditions including migraine, vasomotor headaches, chronic tinnitus, and psychogenic pain attacks found that DSIP administration significantly lowered pain levels in six out of seven patients. Treatment consisted of intravenous administration on five consecutive days followed by five injections every 48-72 hours. Notably, patients experienced simultaneous significant reduction in concomitant depressive states alongside pain relief.
DSIP's analgesic mechanisms appear to involve modulation of endogenous opioid systems and direct effects on pain transmission pathways in the spinal cord. The peptide influences substance P and beta-endorphin levels, both critical mediators of pain perception and modulation. Additionally, DSIP affects pain signal transmission through actions on specific neural pathways and receptors in pain-processing regions of the central nervous system.
The peptide shows particular promise for neuropathic pain caused by nerve damage, a condition often resistant to conventional medications. DSIP helps alleviate symptoms of fibromyalgia, characterized by widespread muscle pain and chronic fatigue. The peptide's combined effects on pain perception, sleep quality, and stress reduction provide comprehensive relief for complex chronic pain conditions where multiple factors contribute to symptom severity.
Sources:
- Kastin AJ, et al. "The effects of DSIP on pain threshold during light and dark periods in rats are not naloxone-sensitive." International Journal of Neuroscience. 1987;37(1-2):65-70. https://pubmed.ncbi.nlm.nih.gov/3679693/
- Schneider-Helmert D, Schoenenberger GA. "Therapeutic effects of delta-sleep-inducing peptide (DSIP) in patients with chronic, pronounced pain episodes. A clinical pilot study." European Neurology. 1983;22(6):414-21. https://pubmed.ncbi.nlm.nih.gov/6548970/
- Nakamura A, et al. "Potent antinociceptive effect of centrally administered delta-sleep-inducing peptide (DSIP)." European Journal of Pharmacology. 1988;155(3):247-53. https://pubmed.ncbi.nlm.nih.gov/3208831/
Addiction Recovery and Withdrawal Symptom Management
DSIP demonstrates remarkable efficacy in treating withdrawal symptoms from both opiates and alcohol, offering a novel physiologically-based approach to addiction recovery. The hypothesis for this therapeutic use stems from DSIP's agonistic activity on opiate receptors, which may help counteract the neurochemical disruptions caused by chronic substance use.
Clinical trials involving 107 inpatients with alcohol (n=47) or opiate (n=60) withdrawal symptoms showed that DSIP administration produced rapid and substantial symptom relief. Of evaluable patients, 97% of opiate-dependent individuals and 87% of alcohol-dependent patients experienced marked improvement or complete disappearance of clinical symptoms following DSIP treatment. Physical withdrawal symptoms including tremors, sweating, muscle cramps, and overall malaise improved rapidly, typically within hours to days of treatment initiation.
A separate study of 67 patients (28 alcoholics and 39 opiate addicts) receiving DSIP as sole treatment for withdrawal symptoms found beneficial effects in 48 of 49 evaluable patients (98%). The treatment produced immediate onset of action with good and lasting suspension of somatic symptoms. Anxiety symptoms resolved more slowly but typically improved within several hours. Importantly, no major side effects occurred during treatment, with only occasional headaches reported by a small number of patients.
The peptide's mechanisms in addiction treatment appear multifaceted. DSIP modulates neurotransmitter systems disrupted by chronic substance use, including dopaminergic, GABAergic, and opioidergic pathways. By helping to restore neurochemical balance, DSIP alleviates the dysphoria, anxiety, and physical discomfort that drive continued substance use during withdrawal. The peptide's stress-reducing properties and improvement of sleep quality provide additional support during the critical early recovery period.
Notably, DSIP treatment required more prolonged administration and higher numbers of injections for opiate-dependent patients compared to alcoholics, reflecting the greater severity and complexity of opiate withdrawal. However, tolerance to DSIP treatment remained good throughout extended administration periods, without development of dependency or abuse potential. This safety profile makes DSIP particularly valuable as an adjunct therapy in comprehensive addiction treatment programs.
Sources:
- Schneider-Helmert D, Schoenenberger GA. "DSIP in the treatment of withdrawal syndromes from alcohol and opiates." European Neurology. 1984;23(5):364-8. https://pubmed.ncbi.nlm.nih.gov/6548969/
- Schneider-Helmert D, Schoenenberger GA. "Successful treatment of withdrawal symptoms with delta sleep-inducing peptide, a neuropeptide with potential agonistic activity on opiate receptors." European Neurology. 1983;22(5):346-51. https://pubmed.ncbi.nlm.nih.gov/6328354/
Neurological Recovery and Neuroprotection
DSIP demonstrates significant neuroprotective properties and facilitates neurological recovery following injury. Research on focal stroke models shows that intranasal DSIP administration accelerates motor function recovery, offering therapeutic potential for stroke rehabilitation and neurological restoration.
A study examining DSIP effects in rats with middle cerebral artery occlusion (MCAO)-induced stroke found that combined preventive and therapeutic DSIP treatment (60 minutes before occlusion and for 7 days post-reperfusion at 120 μg/kg) led to significantly improved motor coordination and balance recovery. While brain infarction size was slightly smaller in DSIP-treated animals compared to controls (though not statistically significant), motor performance in rotarod testing showed significant improvement, indicating enhanced functional recovery independent of lesion size reduction.
The mechanisms underlying DSIP's neuroprotective effects involve multiple pathways. The peptide demonstrates potent antioxidant activity through upregulation of enzymatic antioxidant systems. Research shows DSIP increases expression of genes encoding superoxide dismutase 1 (SOD1) and glutathione peroxidase 1 (GPX1) in brain tissue and blood cells, enzymes whose expression typically decreases during physiological aging. DSIP treatment reduces accumulation of lipid peroxidation products and increases levels of reduced glutathione, providing comprehensive protection against oxidative damage.
Studies on brain mitochondrial function reveal that DSIP administration reduces signs of oxidative stress in neural tissue. This mitochondrial protection is critical for maintaining cellular energy metabolism and preventing the cascade of events leading to neuronal death following injury or during neurodegenerative processes. The peptide's effects on brain metabolism suggest potential therapeutic applications in conditions such as Alzheimer's disease and Parkinson's disease, though specific research in these areas remains limited.
DSIP's influence on sleep quality and circadian rhythms contributes indirectly to neuroprotection. Deep sleep phases facilitated by DSIP are essential for clearance of metabolic waste products from the brain through the glymphatic system, removal of neurotoxic proteins, and consolidation of memories and learned behaviors. Enhanced sleep quality supports overall brain health and may slow age-related cognitive decline.
The peptide's applications in clinical neurology extend to treatment of chemotherapy-induced neurological impairment. A study of children aged 3-16 years who received a 10-day course of DSIP (marketed as Deltaran) following antiblastic therapy showed that the treatment reduced chemotherapy-induced impairment in bioelectrical brain activity in 9 out of 10 children, demonstrating DSIP's potential for mitigating treatment-related neurological side effects.
Sources:
Hormonal Regulation and Growth Hormone Modulation
DSIP influences the secretion of several anterior pituitary hormones, demonstrating complex neuroendocrine regulatory functions. Research indicates the peptide affects growth hormone (GH), luteinizing hormone (LH), adrenocorticotropic hormone (ACTH), and potentially other hormones through actions at both hypothalamic and pituitary levels.
Studies in ovariectomized rats show that intraventricular injection of DSIP induces significant elevation of growth hormone levels within 30 minutes, with effects persisting for 120 minutes following administration. The response is dose-dependent, with minimal effective doses as low as 0.1 micrograms and linear log-dose increases up to 10 micrograms. This GH-releasing effect appears mediated through hypothalamic mechanisms involving dopaminergic pathways, as it can be blocked by pretreatment with pimozide (a dopamine receptor blocker).
In vitro studies using dispersed, cultured pituitary cells demonstrate that DSIP also exerts direct effects on the pituitary gland, producing dose-related increases in GH release. However, the direct pituitary effects show a bell-shaped dose-response curve, suggesting complex receptor interactions. The physiological significance of DSIP's effects on GH secretion is supported by research demonstrating its role in sleep-related growth hormone release.
Evidence from sleep-deprived rats indicates that endogenous DSIP plays a crucial role in the sleep-associated surge of growth hormone. When rats were sleep-deprived for 4 hours, significant increases in both slow-wave sleep and plasma GH levels occurred upon recovery. These increases were completely blocked by microinjection of DSIP antiserum into the third cerebral ventricle, while control animals receiving normal rabbit serum maintained the expected increases. This suggests DSIP functions as a physiological mediator linking sleep, particularly slow-wave sleep, to growth hormone secretion.
DSIP also influences luteinizing hormone (LH) secretion, though effects on follicle-stimulating hormone (FSH) remain unchanged. Intraventricular injection of DSIP caused significant elevation of LH levels within 30 minutes, with values remaining elevated for 2 hours. The minimal effective dose was 1 microgram, and effects were enhanced in animals pretreated with estradiol to lower baseline LH levels. These effects appear mediated primarily through hypothalamic actions rather than direct pituitary stimulation.
Human studies examining DSIP's effects on hormonal secretion have yielded mixed results. While some research shows DSIP administration reduces ACTH-like immunoreactivity in plasma for at least 3 hours, other studies in healthy women found no effects on GH or prolactin secretion at doses known to modify ECG patterns. These discrepancies may reflect species differences, gender-specific responses, or variations in dosing protocols and administration routes.
Sources:
- Iyer KS, McCann SM. "Delta sleep-inducing peptide (DSIP) stimulates growth hormone (GH) release in the rat by hypothalamic and pituitary actions." Peptides. 1987;8(1):45-8. https://pubmed.ncbi.nlm.nih.gov/3575154/
- Obál F Jr, et al. "Evidence for a role of delta sleep-inducing peptide in slow-wave sleep and sleep-related growth hormone release in the rat." Neuroendocrinology. 1987;45(5):393-8. https://pubmed.ncbi.nlm.nih.gov/3035345/
- Laakmann G, et al. "Delta sleep-inducing peptide administration does not influence growth hormone and prolactin secretion in normal women." Experimental and Clinical Endocrinology. 1993;101(4):216-21. https://pubmed.ncbi.nlm.nih.gov/8475226/
Anti-Aging and Geroprotective Effects
DSIP demonstrates remarkable geroprotective (anti-aging) properties in long-term animal studies, extending maximum lifespan, reducing biomarkers of aging, and significantly decreasing spontaneous tumor development. Research shows that chronic DSIP administration produces comprehensive anti-aging effects through multiple complementary mechanisms.
A landmark study examining female Swiss-derived SHR mice receiving monthly 5-day courses of DSIP (100 μg/kg) from age 3 months until natural death revealed significant age-delaying effects. While mean lifespan was not significantly altered, the treatment increased lifespan of the longest-lived 10% of survivors by 17.1% and maximum lifespan by 24.1% compared to controls. These results indicate DSIP slows the aging rate in the population rather than simply reducing early mortality.
DSIP treatment produced multiple beneficial effects on aging biomarkers. The peptide slowed age-related cessation of estrous function in female mice, maintaining reproductive cycling longer than controls. Treatment decreased chromosome aberration frequency in bone marrow cells by 22.6%, indicating protection of genetic integrity during aging. Additionally, DSIP-treated mice maintained higher levels of physical activity, with the longest-lived animals showing significantly greater vertical exploration behavior in behavioral tests throughout their extended lifespans.
The most striking finding was DSIP's profound effect on spontaneous tumor development. Long-term treatment decreased total spontaneous tumor incidence by 2.6-fold compared to controls, with particularly marked reductions in mammary carcinomas and leukemias. This anticarcinogenic effect represents a major component of DSIP's overall geroprotective action, as cancer is a primary cause of mortality in aging populations. The mechanisms underlying this tumor prevention likely involve DSIP's antioxidant effects, immune modulation, and influences on cellular proliferation and apoptosis.
DSIP's antioxidant mechanisms are central to its geroprotective effects. The peptide stimulates activity of key antioxidant enzymes including superoxide dismutase, catalase, glutathione peroxidase, and glutathione reductase, particularly in aged animals where these protective systems typically decline. DSIP increases expression of genes encoding SOD1 and GPX1 in brain and blood cells across various age groups (2-24 months in rats). The peptide reduces accumulation of lipid peroxidation products, stabilizes cellular membranes, and increases levels of reduced glutathione, providing comprehensive protection against oxidative damage that accumulates with aging.
Additional mechanisms contributing to DSIP's anti-aging effects include stimulation of protein biosynthesis, maintenance of mitochondrial function, and modulation of inflammatory responses. The peptide's effects on sleep quality, stress resistance, and hormonal balance create a favorable physiological environment for healthy aging. DSIP's influence on melatonin secretion may also contribute to geroprotective effects, as melatonin possesses well-documented antioxidant and anti-aging properties.
Sources:
- Popovich IG, et al. "Effect of delta-sleep inducing peptide-containing preparation Deltaran on biomarkers of aging, life span and spontaneous tumor incidence in female SHR mice." Mechanisms of Ageing and Development. 2003;124(6):721-31. https://pubmed.ncbi.nlm.nih.gov/12782416/
- Bondarenko TI, et al. "Mechanism of geroprotective action of delta-sleep inducing peptide." Advances in Gerontology. 2012;2(1):8-13. https://doi.org/10.1134/S2079057011040035
- Popovich IG, et al. "[Effect of delta-sleep inducing peptide preparation Deltaran on longevity, physiological functions, and carcinogenesis in mice]." Advances in Gerontology. 2009;22(3):449-59. https://pubmed.ncbi.nlm.nih.gov/20405733/